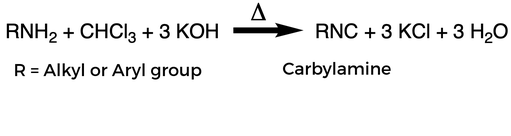
The carbylamine test is a test used to determine whether an organic compound contains an aliphatic or aromatic primary amine group. A primary amine has 2 hydrogens, a secondary amine has 1 hydrogen, and a tertiary amine has no hydrogens. In this test, the unknown compound is reacted with chloroform and potassium hydroxide in the presence of heat. If a carbylamine forms, it will smell bad, although it is toxic so do not purposefully smell the vapors.
The reaction is as follows:
Materials
Test tubes
Test tube rack
Chloroform
Potassium hydroxide
Ice
Compound to be tested
Safety
Do not expose yourself to the vapors while performing the carbylamine test. Carbylamine is highly toxic and should be destroyed immediately after the test. To do this, cool the test tube and carefully add an excess of concentrated hydrochloric acid. After the addition of HCl, put the solution in the correct waste container and do not put the solution down the sink. Chloroform is harmful. Potassium hydroxide is corrosive.
Procedure
Put 2-3 drops of the compound in a test tube.
Add 2-3 drops of chloroform followed by addition of an equal volume of 0.5 M alcoholic potassium hydroxide solution.
Heat the contents gently.
An obnoxious smell of carbylamine confirms the presence of the primary amino group in the compound.
Do not inhale the vapors. Destroy the product immediately by adding concentrated hydrochloric acid and flush it into the sink.
References:
- NCERT Lab Manual for Functional Group tests