卢卡斯test is a test to determine if an organic compound contains a primary, secondary or tertiary alcohol. The Lucas reagent contains zinc chloride and concentrated hydrochloric acid. This reagent reacts with primary, secondary and tertiary alcohols at different rates and the reaction can take between 10 minutes to several days. Alcohols are soluble in Lucas reagent, but the alkyl halides which form from the reaction are not soluble. Therefore, two layers form in the reaction medium. How the layers separate can indicate the type of reaction as shown below.
Primary alcohols: The layers do not separate
Secondary alcohols: The layers separate within 1-5 minutes
Tertiary alcohols: the layers separate immediately
It is important to note that the Lucas test only works if the alcohol is soluble in the Lucas reagent since the result of the test is determined by the separation of the layers.
Example reactions:
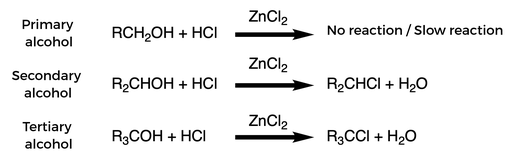
材料
Test tubes
Test tube rack
Lucas reagent
Compound to be tested
Safety
Lucas reagent is highly toxic and corrosive. The vapors should not be inhaled.
Procedure
Put 1 mL of the compound in a test tube.
Add 10 mL of Lucas reagent.
Shake well and note the time for the separation of two distinct layers. This will indicate the type of alcohol present in the organic compound.
References:
- NCERT Lab Manual for Functional Group tests