Depending on its composition,mattercan be classified into several categories. Most matter around us consists of mixtures of pure substances.
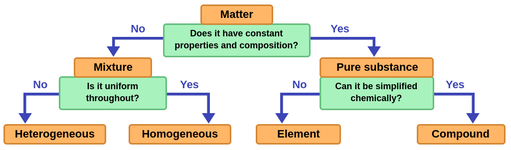
Figure 1.The organization of matter
Apure substanceis a matter that has a constant composition. All samples of a pure substance have precisely the same makeup and properties.
Anelementis a type of substance that cannot be broken into a simpler substance by a chemical change. Each element is composed of only one kind of atom. Gold, copper, hydrogen, and iron are some familiar examples of an element.
Two or more different kinds of elements can combine to make up acompound。例如,食盐是一个复合的缺点ists of two elements: sodium and chloride.
The combination of different substances createsmixtures。Mixtures that are uniform throughout are ahomogeneous mixture, also called a solution. For example; saltwater, the atmosphere, wine, and brass.
A mixture that varies in composition, properties and appearance throughout is aheterogeneous mixture。An oil and water mixture is a heterogeneous mixture because its composition is not uniform throughout. Rocks, sands, and granite are some examples of heterogeneous mixtures.
Each substance in the mixture retains its chemical identity. Using physical methods, we can separate the mixture into its pure substance constituents.