The phase diagram is a graph that represents the phases of a substance as a function of temperature and pressure. The diagram shows the phase-transition temperatures (melting points, sublimation points, boiling points) at different pressures.
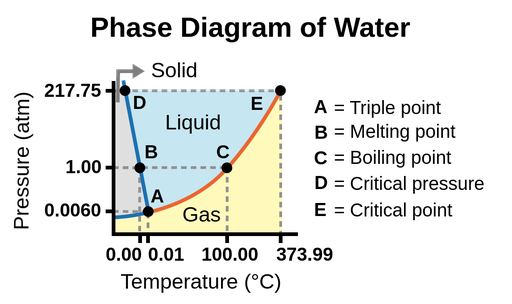
Figure 1.Phase diagram of water. The pressure and temperature axes are not drawn to scale.
- Triple point: Point where all three phases coexist in equilibrium. At pressure lower than the triple point, water cannot exist as a liquid, regardless of the temperature.
- Melting point: The temperature to melt ice.
- Boiling point: The temperature to evaporate water.
- Critical pressure: The pressure required to liquefy a gas at its critical temperature.
- Critical point: Critical temperature and critical pressure of a substance. Beyond the critical point, the physical properties of water are intermediate between gaseous and liquid states. This state is called supercritical fluid.
我们可以使用的相图确定cal state of a substance under specified conditions of pressure and temperature. For example, at 15 atm and -15oC, we can see that water exist in the solid state.
The solid/liquid boundary line has a negative slope, which means that the melting point of ice decreases as the external pressure increases. While the liquid/gas boundary line has a positive slope. This means the boiling point of water increase as the external pressure increases.