Pigments absorb certain wavelengths oflightand reflect others, but why are some molecules colorful and others not?
要理解这一点,我们必须深入研究基金amental structure of atoms. Electrons, just like photons, have both wave and particle nature. The location of an electron inside a molecule can be described as a standing wave, a so-called molecular orbital. You can imagine these molecular orbitals like clouds around the atomic nucleus. The density of the cloud represents the probability of the electron being in a certain position inside the orbital. Each orbital has a different energy level. The orbitals close to the atomic nucleus are lower in energy and are, therefore, more favorable.
The outermost electrons are in the highest energy orbitals and are the most interesting ones. They can be excited by electromagnetic waves. If the energy of a wave matches the energy gap between the highest occupied molecular orbital and the lowest unoccupied molecular orbital, the electron is blasted into the higher energy level and the energy of the electromagnetic wave is absorbed.
Most organic molecules only absorb high energy UV light because the energy gap between the molecular orbitals is quite large. DNA, for example, absorbs wavelengths of 260 nm. Proteins have an absorption peak at 280 nm, hence the ratio of absorbance at 260 nm relative to 280 nm is a simple measurement for the purity of a DNA extract.
In pigments, the energy gap is small enough for visible light to be absorbed. Pigment molecules often contain vast conjugated systems. These systems of connected molecular orbitals with delocalized electrons lower the overall energy of the molecules and decrease the energy gaps between molecular orbitals. Conjugated systems are identified by the presence of alternating single and multiple bonds and lone pairs of electrons (i.e. in oxygen or nitrogen).
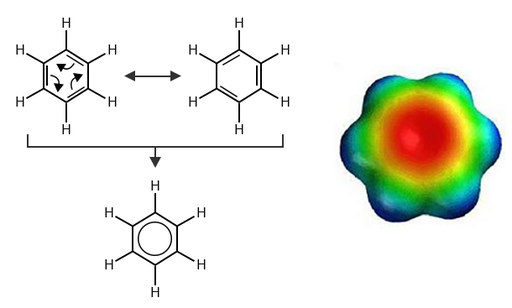
The image above shows a very simple conjugated system. The alternating double bonds can be drawn in either position. In reality, the electrons are delocalized and are spread all over the molecule. The colorful heat map pictured on the right symbolizes the distribution of the electrons over the molecule. Red refers to the maximum electron density.
The more atoms that are part of a conjugated system, the lower the energy gaps are between the molecular orbitals. This is the main reason why pigments are colorful. In addition, often, pigment contains a metal atom that helps shift the energy levels of the molecular orbitals. Chlorophyll and Haeme, the red pigment in our blood, both contain a porphyrin ring with a metal atom in the center.
Chlorophyll can be found in allphotosynthesizingplants and algae. It absorbs light between 400-500 nm and 600-700 nm, depending on the structural groups attached to the porphyrin ring. As you can easily guess from its structure below; chlorophyll is hydrophobic.
