Periodic Table (Principles): Get the table organized in time Virtual Lab
Help Dr. One get the periodic table ready in time! By directly observing the elements’ characteristics, testing their flame color, and investigating trends in atomic properties, your mission is to figure out where a number of fallen out elements belong.

Try Labster's 30-Day All-Access Trial for Free
Discover one of our 200+ simulations today
- Description
- Features
About the Periodic Table (Principles): Get the table organized in time Virtual Lab
Light up the elements! In this simulation, you will learn all the fundamental ins and outs of the periodic table. Find out which elements have metallic characteristics, perform a flame test, and explore trends in atomic properties among the main groups and periods.
A broken table
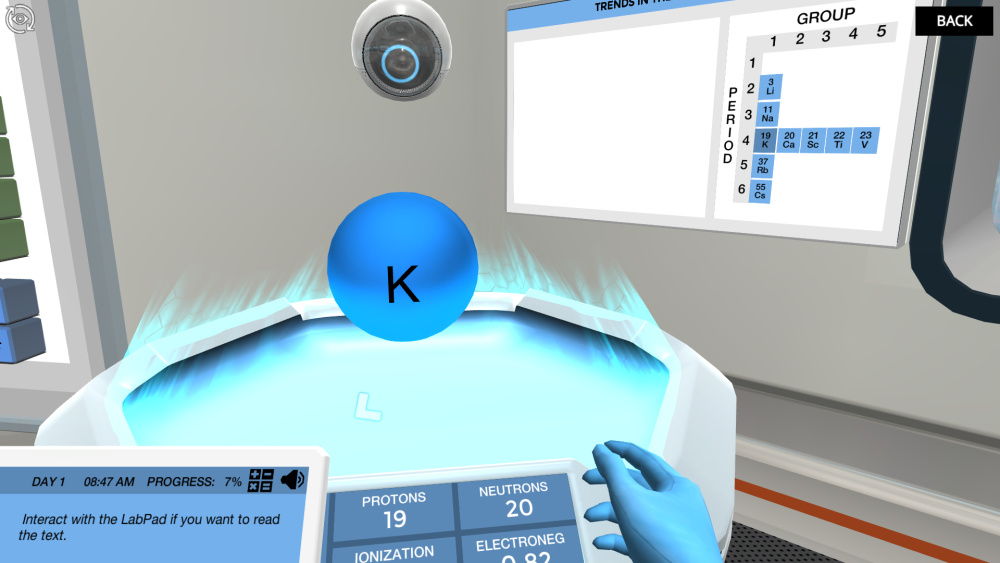
The periodic table is an amazing tool for getting an overview of all the elements in existence, even the elements that only exist when we create them ourselves. But the table in this virtual lab lab is broken; some of the elements have fallen out, and it’s your mission to figure out where they belong by investigating their characteristics and properties.
Flame color test and atomic properties
To sort the first few elements in the table you can observe their flame colors. You may need to use your knowledge about element classes as well to get the full overview.
There are surprisingly many atomic properties for which trends can be observed via the periodic table. Moving on to the virtual holo-table, you will investigate these trends and deduct how they behave when you move within and between periods and groups of the table. You will need this knowledge to assign the most tricky fallen-out elements.
Become the teacher
At the end of the simulation, a special visitor will be coming by the lab, and she needs you to help her understand the basics of the periodic table.
Will you be able to grasp the periodic table well enough to explain it to the visitor?
Help Dr. One get the periodic table ready in time! By directly observing the elements’ characteristics, testing their flame color, and investigating trends in atomic properties, your mission is to figure out where a number of fallen out elements belong.
At the end of this simulation, you will be able to...
- Describe the structure and organization of the periodic table
- 基于他们的locat分类元素的一个家庭ion in the periodic table
- Distinguish metals from other element classes based on typical characteristics
- Use the flame color test to identify metals based on their position in the periodic table
- Relate valence electrons and oxidation state of a main group element to its position in the periodic table
- Describe the main trends among groups and periods for atomic properties
- Explain the causes on the atomic level for the main trends among groups and periods concerning atomic radii, ionization energy and electronegativity
Length: 38 Minutes
Accessibility mode: Available
Languages: English (United States)
HS-PS1-1, HS-PS1-2
Chemistry 3.1 Periodic table
Chemistry 3.2 Periodic trends
1.7 Periodic Trends
1.8 Valence Electrons and Ionic Compounds